Why Is a Bomb Calorimeter at Constant Volume
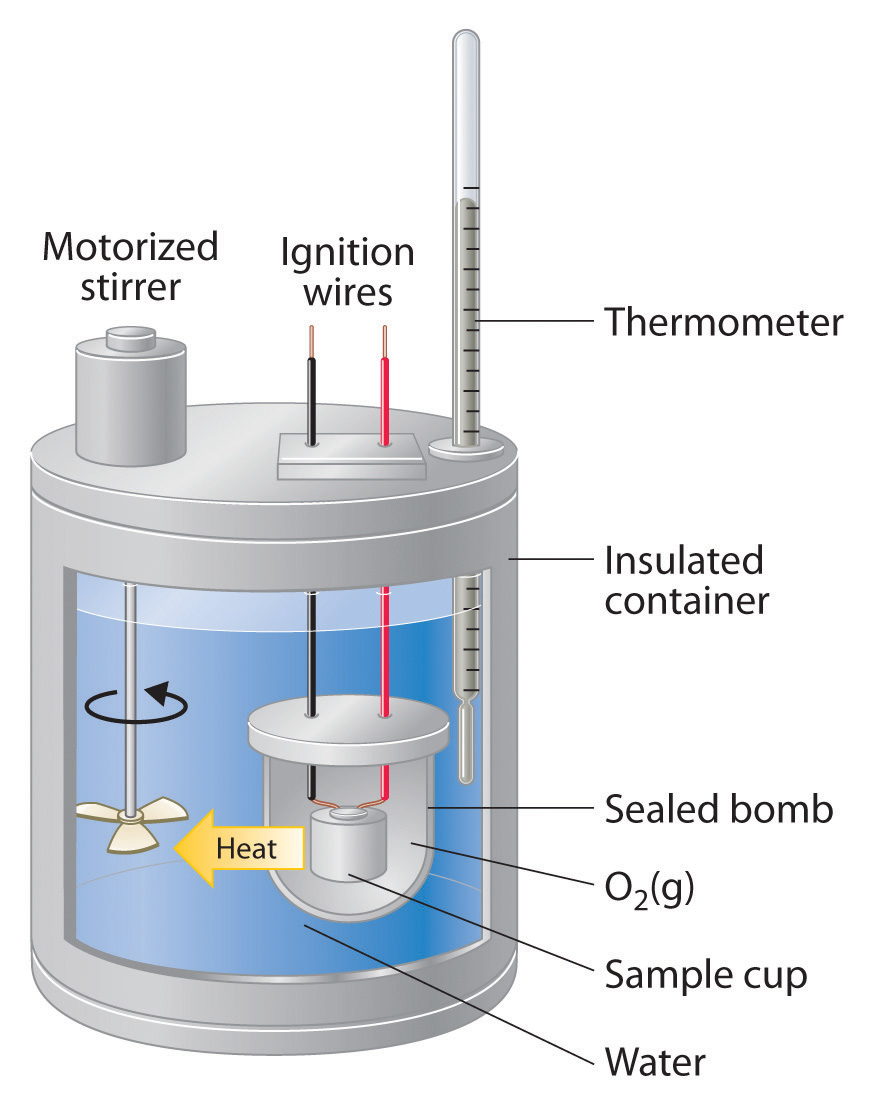
The bomb calorimeter consist primarily of the sample oxygen the stainless steel bomb and water. A bomb calorimeter is an instrument used to measure the amount of heat generated in the combustion of a solid or liquid substance operating at constant volume.

Thermochemistry Constant Volume Calorimeter Bomb Calorimeter Youtube
It records temperature changes continuously with respect to the atmosphere because it is open to the atmosphere thus making it a constant pressure calorimeter.

. The bomb calorimeter is an instrument used to measure the heat of reaction at a fixed volume and the measured heat which is called the change of internal energy ΔE. A bomb calorimeter or a constant volume calorimeter is a device often used to determine the heat of combustion of fuels and the energy content of foods. A bomb calorimeter or a constant volume calorimeter is a device often used to determine the heat of combustion of fuels and the energy content of foods.
It is a constant-volume type calorimeter that measures the heat of a particular reaction or measures the calorific value of the fuels. In a constant volume calorimeter the system is sealed or isolated from its surroundings which accounts for why its volume is fixed and there is no volume-pressure work done. Bomb calorimeters are built in such a way that they can withstand the large pressure produced within the calorimeter due to.
Why Is A Coffee Cup Calorimeter A Constant Pressure Calorimeter. Working of Bomb Calorimeter. Why Is A Bomb Calorimeter Used Instead Of A Coffee Cup.
At constant volume this is equal to qV the heat of reaction. In this technique a sample is burned under constant volume in a device called a bomb calorimeter. It is a constant-volume type calorimeter that measures the heat of a particular reaction or measures the calorific value of the fuels.
Why use a bomb calorimeter instead of a coffee cup. The dewar prevents heat flow from the calorimeter to the rest of the universe ie qcalorimeter 0 Since the bomb is made from stainless steel the combustion reaction occurs at constant volume and there is no work ie wcalorimeter -òp dV 0. In which instance would a bomb calorimeter be more useful than a coffee cup calorimeter.
A bomb calorimeter is an instrument used to measure the heat released from combusting a specific amount of biomass sample and it calculates the HHV of this biomass fuel. The calorimeter has its own heat capacity which must be accounted for when doing calculations. Calorimetry is the process of measuring the amount of heat released or absorbed during a chemical reaction.
What is calorimetry and how does it work. A Bomb calorimeter is a type of constant-volume calorimeter used in measuring the heat of combustion of a particular reaction. A bomb calorimeter is a constant volume calorimeter constant volume is isochoric.
Click to see full answer. Calorimetry is simply the measurement of heat. The heat can be determined from the temperature change ΔT and the heat capacity of the calorimeter Ccal.
A bomb calorimeter is used to measure the change in internal energy ΔU Δ U of a reaction. The amount of heat released in the reaction can be calculated using the equation q -CΔT where C is the heat capacity of the calorimeter and ΔT is the. Gases and reaction conditions are measured for by a bomb calorimeter.
In each testing about one gram of sample fuel is ground and condensed to fit in a capsule for combustion in the bomb. A constant-pressure calorimeter allows volume changes and measures enthalpy. Top 2 posts Page 1 of 1 Return to Heat Capacities Calorimeters Calorimetry Calculations Jump to Who is online.
Equally a bomb calorimeter can not be used to fire gas samples. It is a constant-volume type calorimeter that measures the heat of a particular reaction or measures the calorific value of the fuels. Bomb calorimeters are designed to operate at constant volume such that the volume of the reaction is not allowed to change Δ V 0.
A bomb calorimeter structure consists of the following. Bomb calorimeters are built in such a way that they can withstand the large pressure produced within the calorimeter due to the reaction or burning of fuel. In chemistry the changes of heat of a reaction can be measured at fixed pressure or volume.
Bomb calorimeters have to withstand the large pressure within the calorimeter as the reaction is being measured which might be. The Bomb Calorimeter Model-IKA C2000 was used to measure the cross calorific value of the solid and liquid samples. A bomb calorimeter is used to measure heat flow for solids with low to high temperature reactions.
A constant-volume bomb calorimeter is a fixed volume and therefore pressure changes and measures q v. A bomb calorimeter orimeter is used to burn samples while maintaining constant volumes. Thermometer Ignition wires heat sample Stirrer In an experiment a 04418 g sample of pyrene C16H10 is burned completely in a bomb calorimeter.
By means of bomb calorimetry and respirometry Jensen 131 determined the population density and various energy categories for the slug Arion ater in a Danish beech stand from May to October. A calorimeter is comprised of many. Steel bomb which contains the reactants.
Constant Volume bomb calorimetry is used to measure the heat of a reaction while holding volume constant and resisting large amounts of pressure. Ignition wires heat sample Stirrel In an experiment a 03449 g sample of heptadecanoic acid C17H3402 is burned completely in a bomb calorimeter. So the heat measured by such an instrument is equivalent to the change in internal energy or ΔU.
Therefore the work done is zero and the heat qv measured using a bomb calorimeter is equivalent to the change in internal energy of the reaction. The bomb calorimeter is a type of constant-volume calorimeter used to measure. Calorimetry used for thermonuclear reactions such as bombing the coffee cups could melt the cup.
Water bath in which the bomb is submerged. A bomb calorimeter or a constant volume calorimeter is a device often used to determine the heat of combustion of fuels and the energy content of foods. Thermometer Ignition wires heat Sample Stirrer in an experiment a 05549 9 sample of nonanedioic acid C9H1604 is burned completely in a bomb calorimeter.
Constant-volume calorimetry is used to measure the change in internal energy ΔE for a combustion reaction. Bomb calorimeters can not do a chemical solution reaction because the commencement of the reaction can not be controlled triggered.

Measuring Energy At Constant Volume Using A Bomb Calorimeter Youtube

Bomb Calorimeter Vector Illustration Labeled Educational Stock Vector Royalty Free 1607802400
6 5 Constant Volume Calorimetry Measuring Du For Chemical Reactions Chemistry Libretexts
Comments
Post a Comment